Why you need a Third Party Audit
The type of audit altogether with the reason is further distinguished and categorized. This is done with the terms “1st party”, “2nd party” and “3rd party” audit, whereas the latter is conducted by independent, qualified and experienced auditors. And the “3rd party” audit aims at investigating whether the auditee comply with concerning standards and regulations.
What’s more, auditors from the “3rd party” organization usually exhibit high-level GMP execution ability and professional experience. He/she has a good sense of deficiency depicting, grading and evaluating. And the audit process is well arranged and organized, the items are systematic and structured. Besides, he/she possesses strong follow-up of deficiencies, i.e. systematic failure investigation and strict follow-up on suitable CAPA measures when compared with other auditors.
-
The meaning for Third Party Audit
Nowadays pharmaceutical companies face ever-changing market forces and statutory regulations. Hence, they must demonstrate increasingly systematical risk management and consistent GMP compliance. The “3rd party” audit department offers support and guidance in pharmaceutical quality management, assisting the auditee enhance their market competence.
The professional “3rd party” audit can also efficiently save clients’ time and resources, providing them superior suppliers and products through organized auditee management.
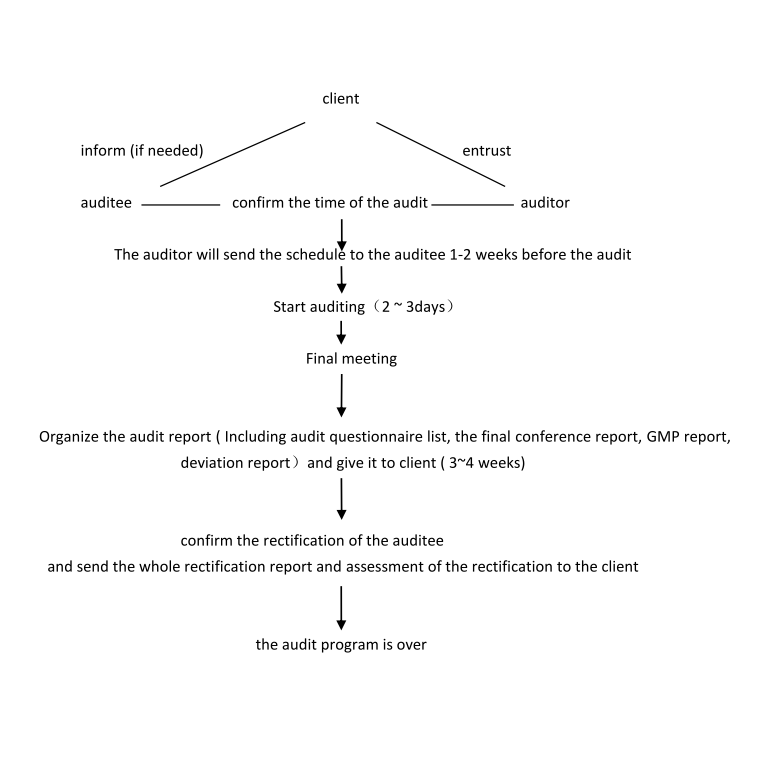
How to perform an audit
Usually, we will send our questionnaire one or two weeks before the audit to the auditee that he can prepare the facility for the inspection. Our client will receive the same questionnaire at the same time. A few days after the audit,ourclient will receive a document regarding the final discussion at the factory, which includes a preliminarydeviation report. The GMP report, which states, if the facility adheres to the European GMP requirements, the final deviation report and the finalized questionnaire, which identifies the documents we have assessed, will be sent in due course.